 When mass defect is in atomic mass unit ( u), the calculated binding energy will be in MeV (by multiplying by 931.5).įor example, the binding energy of carbon-12 (nuclide mass = 12.0000 u) nucleus can be calculated:įirst, calculate total mass of nucleons (6 protons and 6 neutrons) The energy released by fusion is three to four times greater than the energy released by fission.When mass defect is in kg, the calculated binding energy will be in J.Mass Defect and Binding Energy Calculation Conversely, when a nucleus/atom is broken apart, its binding energy is absorbed.īinding energy per nucleon equals to the total binding energy of a nucleus divided by the number of nucleons in the nucleus.īinding energy per nucleon varies with nuclide - generally increases up to iron (Fe) and decreases after iron. Nuclear Structure of atomic species changes. The energy output of fusion is several times greater than.  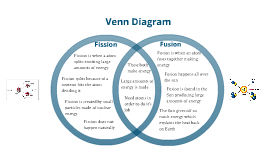
The missing mass is converted into energy. The newly formed atom will contain slightly less mass than the sum of the atoms that were used to create it. Electrons in the atom are responsible for chemical reactions. Nuclear fusion is the opposite of fission, in that it involves the fusing of two or more atoms together to form a new, heavier element. The reason why opposite processes release. In both cases, energy is freed because the mass of the remaining nucleus is smaller than the mass of the reacting nuclei. When a nucleus/atom is formed, its binding energy is released. Nuclear reaction takes place in the atoms nucleus. The method of nuclear fusion is versus that of nuclear fission. Fission splits a heavy element (with a high atomic mass number) into fragments while fusion joins two light elements (with a low atomic mass number), forming a heavier element. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
